Antimicrobial peptide LL-37, belongs to the cathelicidin family of peptides, and this peptide corresponds to the sequence of the first amphipathic alpha-helical peptide isolated from humans. They are small, cationic peptides found in humans and other species. Specifically, they are stored in neutrophil granules as inactive precursors and are released as mature peptides when required and cleaved by neutrophil elastase.
Buy LL-37 is expressed in various cells and tissues such as circulating neutrophils and myeloid bone marrow cells, epithelial cells of the skin, and is also expressed in the gastrointestinal tract, as well as in the epididymis and lungs. Expression was also detected in squamous epithelium of the mouth, tongue, esophagus and in the colonic and bronchial mucosal epithelium.
Moreover, production of buy LL-37 in macrophages is stimulated by vitamin D released by sunlight through the skin. LL-37 plays an important role in the first line of defense against infection and systemic invasion of pathogens at sites of inflammation and wound. It is cytotoxic to both bacterial and normal eukaryotic cells and is significantly resistant to proteolytic degradation in solution. They show a broad spectrum of antimicrobial activity against bacteria, enveloped viruses, and fungi. Along with exerting direct antimicrobial effects, they are also critical in triggering specific defense responses in the host.
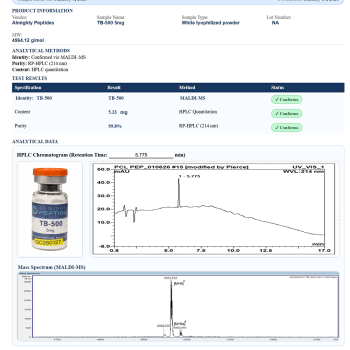
Get buy other products: buy tb-500


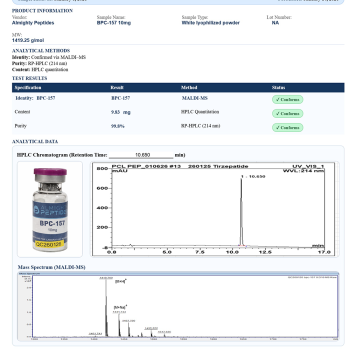
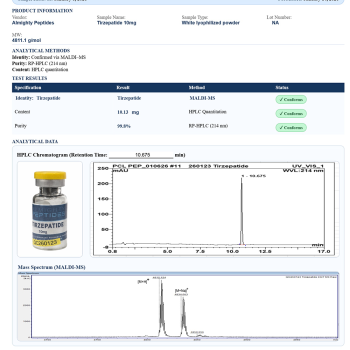

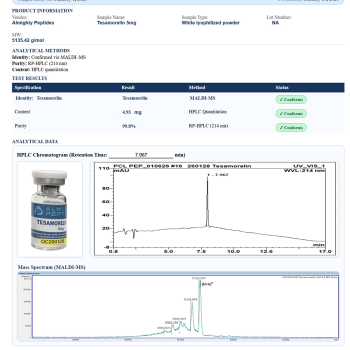




Reviews
There are no reviews yet.